
The diagram above is the solubility curve of a solute, X. Find the amount of X deposited when 500cm3 of a solution of X is cooled from 60oC to 20oC.
Options:A) 0.750 mole
B) 0.950 mole
C) 2.375 moles
D) 4.750 moles
Show Answer
A) Fe3 + (H2O)6
B) FeO.H2O
C) Fe2O3.3H2O
D) Fe3O4.2H22O
Show Answer

The functional group represented in the compound above is
Options:A) alkanol
B) alanal
C) alkanone
D) alanoate
Show Answer
A) Fe is oxidized to Fe3+
B) Fe3+ is oxidized to Fe2+
C) I- is oxidized to I2
D) I- is reduced to I2
E) I- is displacing an electron from Fe3+
Show Answer

The diagram shown above represents the solubility curves of two salts, X and Y, in water, use this diagram to answer the question. If the molar mass of X is 36 g, the number of moles of X dissolved at 343 K is
Options:A) 0.2 moles
B) 0.7 moles
C) 1.5 moles
D) 2.0 moles
E) 3.0 moles
Show Answer
A) make the water colourless
B) remove germs from the water
C) make the water tastedful
D) remove odour from the water
Show Answer
The end products of burning a candle in the atmosphere are water and
Options:A) carbon (II) oxide
B) sulphur (IV) oxide
C) carbon (IV) oxide
D) sulphur (VI) oxide
Show Answer
Which of the following is an example of a chemical change?
Options:A) dissolution of salt in water
B) rusting of iron
C) melting of ice
D) separating a mixture by distillation
Show Answer
A) It is heavy with a high melting point
B) It is malleable and has high density
C) It is strong and light
D) It is hard and ductile
Show Answer
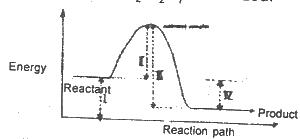
The diagram above shows the reaction path of an exothermic reaction. The heat of reaction is represented by
Options:A) l
B) ll
C) lll
D) lV
Show Answer