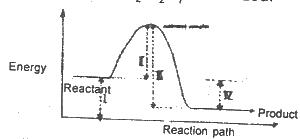
The diagram above shows the reaction path of an exothermic reaction. The heat of reaction is represented by
Options:A) l
B) ll
C) lll
D) lV
Show Answer
The correct answer is D .
A) Mn2+
B) Cl-
C) H2O
D) MnO-4
E) Cl2
Show Answer
The correct answer is D .

The curve depicts titration between
Options:A) Strong acid and strong base
B) Strong acid and weak base
C) Weak acid and weak base
D) Weak acid and strong base
Show Answer
The correct answer is C .
A) number of molecules of the gas
B) density of the gas molecules
C) number of collisions between the gas molecules
D) number of collisions between the gas molecules and the walls of the container
Show Answer
The correct answer is D .
A) Na2SO4
B) Na2S
C) Na2S2O3.5H2O
D) Na2CO3
E) NaHCO3
Show Answer
The correct answer is C .
What mass of water is produced when 8.0g of hydrogen reacts with excess oxygen?
[H = 1, O = 16]
Options:A) 36.0g
B) 8.0g
C) 72.0g
D) 16.0g
Show Answer
The correct answer is C .
A substance that is used as a ripening agent for fruits is
Options:A) ethene
B) propane
C) methane
D) butane
Show Answer
The correct answer is A .
A) cacium
B) magnesium
C) tin
D) copper
Show Answer
The correct answer is C .
A) sodium
B) nitrogen
C) sodium dioxonitrate (III)
D) sodium oxide
Show Answer
The correct answer is C .
A) Au3+
B) Fe3+
C) F-
D) Ca2+
Show Answer
The correct answer is C .