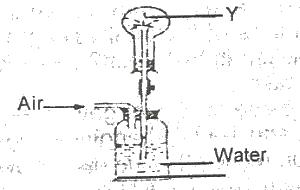
In the diagram above the gas Y could be
Options:A) hydrogen chloride
B) oxygen
C) carbon (IV) oxide
D) chlorine
Show Answer
The correct answer is A .
A) potassium nitrate
B) sand
C) lead nitrate
D) sugar (glucose)
E) starch
Show Answer
The correct answer is B .
A) 2.50 g
B) 2.73 g
C) 3.28 g
D) 4.54 g
Show Answer
The correct answer is C .
A) Acids react with some metals, liberating hydrogen
B) Acids react with carbonates, liberating carbondioxide
C) Acids conduct electricity and are decomposed by the current, liberating hydrogen at the anode
D) Acids have a sour taste
E) Acids change the colour of indicators
Show Answer
The correct answer is C .
8g of CH
A) 3.7dm
B) 11.2dm
C) 22.4dm
D) 33dm
Show Answer
The correct answer is B .
Which transition metal is known for its multiple colorful oxidation states and compounds used in pigments and paints?
Options:A) Silver (Ag)
B) Copper (Cu)
C) Gold (Au)
D) Zinc (Zn)
Show Answer
The correct answer is B .
A) 50, 50
B) 25, 50
C) 50, 25
D) 25, 25
Show Answer
The correct answer is D .
A) Electrovalent
B) Metallic
C) Covalent
D) Dative
Show Answer
The correct answer is A .
Which of the following is a common laboratory indicator for bases?
Options:A) Methyl orange
B) Phenolphthalein
C) Bromothymol blue
D) Litmus
Show Answer
The correct answer is B .
A) butane , propane and kerosene
B) butane , propane and petrol
C) ethane, methane and benzene
D) methane, ethane and propane
Show Answer
The correct answer is D .