The lanthanides and actinides are located in which block of the periodic table?
Options:A) P-block
B) S-block
C) F-block
D) D-block
Show Answer
The correct answer is C .
A) Neutrons 53, Protons 17
B) Neutrons 17, Protons 36
C) Neutrons 19, Protons 17
D) Neutrons 36, Protons 17
Show Answer
The correct answer is C .
A) 0.05 mol -3HCI and 5g powdered CaCO3
B) 0.05 mol -3HCI and 5g lump CaCO3
C) 0.10 mol -3HCI and 5g powdered CaCO3
D) 0.025 mol -3HCI and 5g powdered CaCO3
Show Answer
The correct answer is C .
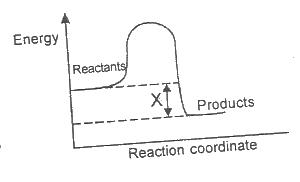
In the diagram above, X is the ``
Options:A) enthalpy
B) enthalpy change
C) activation energy
D) activated complex
Show Answer
The correct answer is B .
A) Over-head cables
B) Duralumin
C) Container for caustic soda
D) Container for nitric acid
Show Answer
The correct answer is D .
A) 6.02 x 1022
B) 5.85 x 1023
C) 6.02 x 1023
D) 5.85 x 1024
Show Answer
The correct answer is A .
The reaction above is an example of
Options:A) nuclear fission
B) nuclear fusion
C) artificial transmutation
D) beta decay
Show Answer
The correct answer is C .
A) deliquescene
B) hygroscopy
C) effervescence
D) efflorescence
Show Answer
The correct answer is D .
A) 52.0g
B) 58.5g
C) 85.5g
D) 88.5g
Show Answer
The correct answer is A .
A) Carbon (IV) oxide
B) Sulphur (Vl) oixide
C) Sulphur (IV) oxide
D) Carbon (ll) oxide
Show Answer
The correct answer is D .