2X(aq) + MnO2(s) + 4H+(aq) → X2(g) + Mn2+(aq) + 2H2O.
The reaction above can be used for the laboratory preparation of all halogens except fluorine because it is?
Options:A) a poisionous gas
B) an oxidizing agent
C) electronegative in nature
D) highly reactive
Show Answer

The set-up above would be useful for determining the amount of
Options:A) oxygen in air
B) water vapour in air
C) CO2 in air
D) arygen in air
Show Answer
A) Pb
B) Ca
C) Na
D) Mg
Show Answer
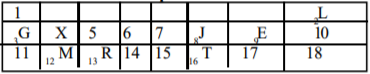
Use the section of the periodic table above to answer this question.
Which of the indicate an alkali metal and a noble gas respectively?
Options:A) M and E
B) G and E
C) R and L
D) G and L
Show Answer
A) 10-10 mol dm-3
B) 10-6 mol dm-3
C) 10-4 mol dm-3
D) 10-2 mol dm-3
Show Answer
A) Na2CO3
B) CaCO3
C) MgCO3
D) ZnCO3
Show Answer

Which of the following compounds will take up the molecules of bromine?
Options:A) l
B) ll
C) lll
D) l and ll
E) l and lll
Show Answer
A) optical isomerism
B) positional isomerism
C) functional isomerism
D) geometrical isomerism
Show Answer
What is the percentage by mass of oxygen in Al2(SO4)3. 2H2O?
[Al = 27, S = 32, H = 1, O = 16]
Options:A) 14.29%
B) 25.39%
C) 50.79%
D) 59.25%
Show Answer
Which of the following alkaline metals react more quickly spontaneously with water?
Options:A) Calcium
B) Beryllium
C) Magnesium
D) Strontium
Show Answer