A) are of low melting and boiling points
B) have weak metallic bonding
C) conduct electricity and heat
D) are very reactive
Show Answer
The correct answer is D .
A) sp 4
B) sp 3
C) sp2
D) sp
E) s
Show Answer
The correct answer is D .
A) deliquescent
B) hygroscopic
C) efflorescent
D) hydrated
E) fluorescent
Show Answer
The correct answer is A .

Use the figure above to answer this question. Which of the following is NOT a monomer?
Options:A) A
B) B
C) C
D) D
Show Answer
The correct answer is A .
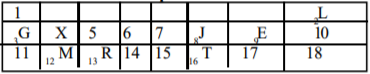
Use the section of the periodic table above to answer this question.
Which of the indicate an alkali metal and a noble gas respectively?
Options:A) M and E
B) G and E
C) R and L
D) G and L
Show Answer
The correct answer is D .
A) Copper
B) Iron
C) Lead
D) Mercury
Show Answer
The correct answer is B .
A) NaHSO4
B) Na2SO3
C) NaHSO3
D) Na2SO4
Show Answer
The correct answer is B .
CH3 - CH2 - C CH
The hybridization in the compound above is
Options:A) Sp2
B) sp
C) sp3 and sp2
D) sp3 and sp
Show Answer
The correct answer is D .
A) the same at the same temperature
B) different at the same temperature
C) the same at different temperature
D) constant
Show Answer
The correct answer is B .
What volume of oxygen is produced from the decomposition of 2 mole of KCIO3 at s.t.p?
[Molar volume of a gas at s.t.p = 22.4 dm3]
Options:A) 22.4 dm3
B) 33.6 dm3
C) 44.8 dm3
D) 67.2 dm3
Show Answer
The correct answer is D .