NH3 + H3O+
→ NH4+ + H2O. It may be deduced from the reaction above that?
Options:A) a redox reaction has occurred
B) H3O+ acts as an oxidizing agent
C) H3O+ acts as an acid
D) water acts as ab acid
Show Answer
The correct answer is C .
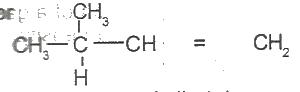
The IUPAC name for
Options:A) 2-methylbut-3-ene
B) 2-methylbut-4-ene
C) 3-methylbut-2-ene
D) 3-methylbut-1-ene
E) 3-methylpent-1-ene
Show Answer
The correct answer is D .
For the reaction NH4NO2
A) 2.24 dm 3
B) 224 dm3
C) 1.12 dm 3
D) 112 dm 3
E) 4.48 dm 3
Show Answer
The correct answer is D .
A) 1
B) 2
C) 3
D) 4
Show Answer
The correct answer is A .
A) carbon (IV) oxide, nitrogen and the rare gases
B) nitrogen and the rare gases
C) carbon (IV) oxide, nitrogen (IV) oxide and the rare gases
D) carbon (IV) oxide
Show Answer
The correct answer is C .
A) Na+
B) Ca2+
C) Fe3+
D) Fe2+
Show Answer
The correct answer is B .
A) Kerosine
B) turpentine
C) a solution of borax in water
D) ammonia solution
Show Answer
The correct answer is C .
A) atoms can neither be created nor destroyed
B) the particles of the same element are exactly alike
C) particles of different elements combine in simple whole number ratio
D) all elemets are made of small indivisible particles
Show Answer
The correct answer is A .
A) CO2
B) SO3
C) CH4
D) H2
Show Answer
The correct answer is A .
A) Copper
B) Mercury
C) Silver
D) Zinc
E) Gold
Show Answer
The correct answer is D .