A) complex
B) neutral
C) basic
D) acidic
Show Answer
The correct answer is B .
A) i, iii and iv
B) iii only
C) ii, iii and i
D) iv only
Show Answer
The correct answer is D .
A) mettylamine and ethanol
B) ethylamine and methanol
C) methanamide and ethanol
D) ethanamide and methanol
Show Answer
The correct answer is D .
A)
B) X-rays
C)
D)
Show Answer
The correct answer is C .
A) Chlorine, aluminium, magnesium, phosphorus, sodium
B) sodium, magnesium, aluminium, phosphorus, chlorine
C) Chlorine, phosphorus, phosphorous, aluminium, magnesium, sodium
D) Sodium, chlorine, phosphorus, magnesium, aluminium
Show Answer
The correct answer is B .
Which of the following are chemical changes?
I. cooking of food
II. digestion of food
III. freezing of water
IV. water is heated up
A) I and II
B) I, II, and III
C) III and IV
D) all of the above
Show Answer
The correct answer is A .
Which of the following types of alkanols undergo oxidation to produce alkanoic acids.I. Primary alkanols
II. Secondary alkanols
III. Tertiary alkanols
Options:A) I, II and III
B) I and II only
C) III only
D) I only
Show Answer
The correct answer is D .
A) electrons
B) Holes and electron
C) Ions
D) Charges
Show Answer
The correct answer is C .
A) 88.71 KJ
B) 85.48 KJ
C) -204.00 KJ
D) -3427.40 KJ
Show Answer
The correct answer is D .
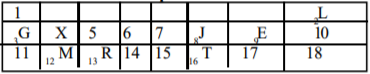
Use the section of the periodic table above to answer this question.
Which of the indicate an alkali metal and a noble gas respectively?
Options:A) M and E
B) G and E
C) R and L
D) G and L
Show Answer
The correct answer is D .