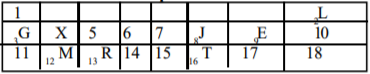
Use the section of the periodic table above to answer this question.
Which of the indicate an alkali metal and a noble gas respectively?
Options:A) M and E
B) G and E
C) R and L
D) G and L
Show Answer
22688Ra → x86Rn + α - particle
What is the value of x in the nuclear reaction above?
Options:A) 220
B) 222
C) 226
D) 227
Show Answer
A) isoprene
B) polythene
C) ethanol
D) benzene
Show Answer
At 25o and zymase as catalyst,
C6H126 → 2C2H5OH + 2CO2 + energy
The reaction above represented by the equation above is useful in the production of
Options:A) propanol
B) butanol
C) methanol
D) ethanol
Show Answer
A) Z is efflorescent
B) Y and Z are efflorescent
C) Y and Z are deliquescent
D) Y and Z are hygroscopic
E) Y is deliquescent
Show Answer
A) H(s)S is reduced to S
B) Fe3+ ions are oxidized by H(s)S
C) Fe3+ ions are oxidized Fe3+ ions
D) Fe3+ ions are reduced to Fe2+ ions
Show Answer
A chemical reaction in which the hydration energy is greater than the lattice energy is referred to as
Options:A) A reversible reaction
B) A spontenous reaction
C) An endothermic reaction
D) An exothermic reaction
Show Answer
At 27°C, 58.5g of sodium chloride is present in 250cm
(molar mass of sodium chloride = 111.0gmol
A) 2.0 moldm
B) 0.25 moldm
C) 1.0 moldm
D) 0.5 moldm
Show Answer
A) The dissolution of NaOH(s) in water is endothermic
B) The heat os solution of NaOH(s) is positive
C) The NaOH(s) gains heat from the surroundings
D) The heat of solution of NaOH(s) is negative
Show Answer
A) Thermal dissociation of ammonium chloride
B) Reaction between an acid and a base
C) Addition of concentrated acid to water
D) Dissolution of sodium metal in water
Show Answer