A) Ca2+
B) Mg2+
C) Zn2+
D) Cu2+
Show Answer
A) ethane, ethene, ethyne and benzene
B) alkanes, alkenes, alkynes and aromatics
C) alkanes, alkenes, alkynes and benzene
D) methane, ethane , propane and butane
Show Answer
A) 3
B) 5
C) 7
D) 10
Show Answer
A sample of a substance containing only C and H burns in excess O2 to yield 4.4g of CO2
And 2.7 g of H2O. The empirical formula of a substance is?(C = 12, O = 16, H = 1)
Options:A) CH3
B) CH2
C) CH4
D) C2H24
Show Answer
Which of the following describes the chemical property of acids?
Options:A) None of the above
B) Acid + XCO
C) Acid + NaOH
D) Acid + X
Show Answer
A) Fractional distillation of crude petroleum will give the folloing hydrocarbon fuels in order of increasing boiling point
B) Butane < petrol < kerosene of polythene.
C) Both but-1-ene and but-1-yne will decolorize bromine readily
D) but-2-ene will react with chlorine to form 2,3-dichlorobutane
E) calcuim carbide will react with water to form any alkyne
Show Answer
2X(aq) + MnO2(s) + 4H+(aq) → X2(g) + Mn2+(aq) + 2H2O.
The reaction above can be used for the laboratory preparation of all halogens except fluorine because it is?
Options:A) a poisionous gas
B) an oxidizing agent
C) electronegative in nature
D) highly reactive
Show Answer
1.1 g of CaCI2 dissolved in 50 cm3 of water caused a rise in temperature of3.4°C. The heat of reaction, ∆H for CaCI2 in kj per mole is?
(Ca = 40, CI = 35.5, specific heat of water is 4.18 jk-1
Options:A) -71.1
B) -4.18
C) +71.1
D) +111.0
Show Answer
A) 5 dm 3
B) 20 dm 3
C) 15 dm 3
D) 10 dm 3
Show Answer
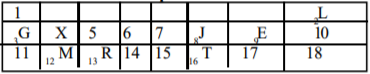
Use the section of the periodic table above to answer this question.
Which of the indicate an alkali metal and a noble gas respectively?
Options:A) M and E
B) G and E
C) R and L
D) G and L
Show Answer