
What is the IUPAC name for the compound?
Options:A) 1-chloro-2-methylprop-2,3-ene
B) 1-chloro-2-methylprop-2-ene
C) 3-chloro-2-methylprop-1-ene
D) 3-chloro-2methylprop-1,2-ene
Show Answer
A) increasing the temperature and decreasing the pressure
B) increasing the pressure and increasing the temperature
C) decreasing the temperature and increasing the pressure
D) decreasing the pressure and decreasing the temperature
Show Answer
Equal volumes of CO,SO2, and H2S were released into a room at the same point and time. Which of the following gives the order of diffusion of the gases to the opposite corner of the room?
(S = 32, C = 12, O = 16, N = 14, H = 1)
Options:A) CO, SO2,NO2,H2S
B) SO2,NO2,H2S,CO
C) CO,H2S,SO2,NO2
D) CO,H2S,NO2,SO2
Show Answer
8g CH4 occupies 11.2 dm3 at s.t.p. What volume would 22 g of CH3CH3 occupy under the same condition?
(C = 12, H = 1)
Options:A) 3.7 dm3
B) 11.2 dm3
C) 22.4 dm3
D) 33.6 dm3
Show Answer
2H
From the equationabove, calculate the volume of unreacted oxygen gas if a mixture of 50cm
A) 85cm
B) 50cm
C) 125cm
D) 55cm
Show Answer
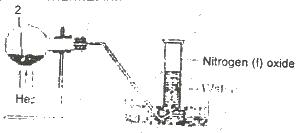
In the experiment above, Z can be
Options:A) a solution of sodium dioxonitrate (lll) and ammonium chloride
B) a solution of lead trioxnitrate (V)
C) a solution of sodium trioxonitrate (V) and ammonium chloride
D) concentrated tetraoxosulphate (VI) acid and sodium trioxonitrate (V)
Show Answer
A) 2SO3(g) ↔ 2SO2(g) + O2(g)
B) 2CO2(g) ↔ 2CO(g) + O2(g)
C) 2H2(g) + O2(g) ↔ 2H2O(g)
D) 2NO(g) ↔ N2(g) + O2(g)
Show Answer
A) K+
B) KNO3-
C) S2-
D) H3O+
Show Answer
Which of the following are mixtures?
I. Petroleum
II. Rubber latex
III. Vulcanizer's solution
IV. Carbon sulphide
Options:A) I, II and III
B) I, II and IV
C) I and II only
D) I and IV
Show Answer
Calculate the percentage composition of oxygen in calcium trioxocarbonate(IV) [Ca=40, C=12, O=16]
Options:A) 16
B) 48
C) 40
D) 12
Show Answer