A) alkyne
B) isoprene
C) n-propene
D) peoprene
Show Answer
The correct answer is B .
A) make the water colourless
B) remove germs from the water
C) make the water tastedful
D) remove odour from the water
Show Answer
The correct answer is B .
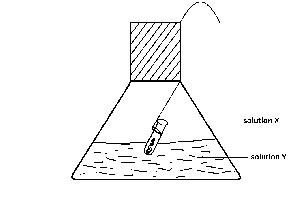
An experiment test of the Law of conservation of Mass is illustrated in the diagram. In practice, the flask is weighed before and after reaction between solutions X and Y. Which of the following pairs of solutions will be unsuitable for the experiments
Options:A) X = hydrochloric acid; Y = silver nitrate
B) X = barium chloride; Y = dilute sulphuric acid
C) X = hydrochloric acid; Y = sodium hydroxide
D) X = hydrochloric acid; Y = lead nitrate
E) X = hydrochloric acid; Y = sodium carbonate
Show Answer
The correct answer is E .
A) dissolution
B) slaking
C) liming
D) mortaring
Show Answer
The correct answer is B .
A) Chlorine gas and hydrogen
B) Oxygen and oxochlorate (I) acid
C) Chlorine gas and oxochlorate (I) acid
D) Hydrochloric acid and oxygen
Show Answer
The correct answer is D .
A) i, iii and iv
B) iii only
C) ii, iii and i
D) iv only
Show Answer
The correct answer is D .
A) HNO3
B) CH3COOH
C) H2SO4
D) HCl
Show Answer
The correct answer is C .
A) 200 g
B) 160 g
C) 100 g
D) 50 g
Show Answer
The correct answer is C .
A) solution
B) suspension
C) precipitate
D) colloid
Show Answer
The correct answer is C .
A) electropositivity decreases from Li to Na to K
B) electronegativity decreases from Li to Na to K
C) the number of the electron shells increases from Li to Na to K
D) the elements are in the same period
Show Answer
The correct answer is C .