A) Salt
B) Dye
C) Mordant
D) Dispersant
Show Answer
The correct answer is C .
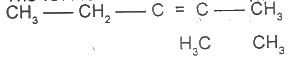
The IUPAC name for the compound is
Options:A) 2,3-dimethylpent-2,3-ene
B) 2, 3- dimethylpent-2-ene
C) 3, 4-dimethylpent-3-ene
D) 3,4-dimethylpentene
E) 3,4-dimethlhept-2-ene
Show Answer
The correct answer is B .
A) Cu (s) + 2HNO 3(aq) → Cu (NO 3) 2(aq) + H 2(g)
B) Cu (s) + 4HNO 3(aq) → Cu (NO 3) 2(aq) + 2H 2O (1) + 2NO 2(g)
C) 3Cu (s) + 8HNO3(aq) → 3Cu (NO3)2(aq) + 4H2O(1) + 2NO(g)
D) 3Cu(s) + 4HNO3(aq) → 3Cu(NO 3) 2(aq) + 2H 2O (1) + 2NO (g)
Show Answer
The correct answer is B .
A) zinc
B) lead
C) iron
D) copper
Show Answer
The correct answer is C .
A) trichloromethane and tetrachloromethane
B) chloroethene and dichloroethene
C) trichloroethene and tetrachloroethene
D) chloroethane and dichloroethene
Show Answer
The correct answer is A .
A) 1s2 2s22p6 3s2 3p6 4s2 3d2
B) 1s2 2s22p6 3s2 3p6 4s2 3d1
C) 1s2 2s22p6 3s2 3p6
D) 1s2 2s22p6 3s2 3p6 4s2
Show Answer
The correct answer is D .

2HCL(ag) + CaCO3(ag) → CaCL2(ag) + H2O(1) + CO
From the reaction above, which of the following curves represents the consumption of calcium trioxocarbonate (IV) as dilute HCL is added to it?
Options:A) L
B) M
C) N
D) P
Show Answer
The correct answer is B .
A) They can be decomposed by heat
B) They react with aqueous AgNO3 to give a white precipiate which is soluble in excess ammonia
C) They explode when in contact with dry ammonia
D) They react with concentrated tetraoxosulphate (IV) acid to give white fumes of chlorine gas
Show Answer
The correct answer is B .
What is the mass percentage of carbon (C) in methane (CH4)? (The molar mass of carbon is approximately 12 g/mol.)
Options:A) 25%
B) 12%
C) 75%
D) 50%
Show Answer
The correct answer is C .
A) sodium is formed at the anode
B) chlorine is formed at the anode
C) sodium does not react with platinum
D) chlorine does not react with platinum
Show Answer
The correct answer is C .