A) 2.00 M aqueous solution of NaOH
B) 0.01 M aqueous solution of NaOH
C) 0.01 M aqueous solution of hexanoic acid
D) 0.01 M aqueous solution of sugar
Show Answer
The correct answer is D .
A) Heat content (enthalphy)
B) Energy of activation
C) Free energy change
D) Equilibrium position
Show Answer
The correct answer is B .
A) 0 oC
B) -100 oC
C) -273 oC
D) -373 oC
Show Answer
The correct answer is C .
An element used in the production of matches is
Options:A) nitrogen
B) aluminium
C) copper
D) sulphur
Show Answer
The correct answer is D .
A) nitrogen
B) hydrogen
C) chlorine
D) sulphur
E) oxide
Show Answer
The correct answer is A .
1.00 g of the oxide of metal X (atomic mass = 64) gave 0.80 g of the metal when reduced with hydrogen.
The formula of the oxide is?
Options:A) X2O
B) XO
C) XO2
D) X2O3
E) X3O4
Show Answer
The correct answer is B .
Which of the following is a common property of non-metals?
Options:A) Exist as solids at room temperature
B) Tend to gain electrons in chemical reactions
C) High thermal conductivity
D) Readily form cations in chemical reactions
Show Answer
The correct answer is B .
The shapes of water, ammonia, carbon (iv) oxide and methane are respectively
Options:A) linear, tetrahedral, angular and pyramidal
B) angular, pyramidal, linear and tetrahedral
C) pyramidal, linear, tetrahedral and angular
D) tetrahedral, angular, pyramidal and linear
Show Answer
The correct answer is B .
A) an increase in the tendency to lose electron
B) an increase in chemical reactivity
C) a decrease in electropositivity
D) a decrease in covalency in bond formation
Show Answer
The correct answer is B .
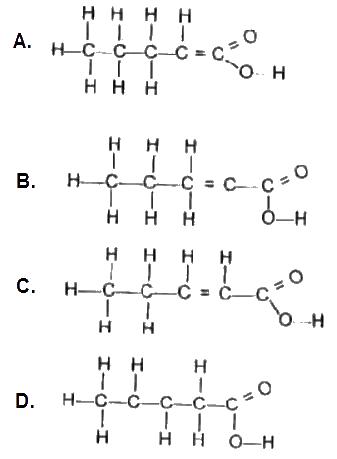
Use the options above to answer this question. Which is the structural formula for pent-2-enoic acid?
Options:A) A
B) B
C) C
D) D
Show Answer
The correct answer is C .