A) Cu (s) + 2HNO 3(aq) → Cu (NO 3) 2(aq) + H 2(g)
B) Cu (s) + 4HNO 3(aq) → Cu (NO 3) 2(aq) + 2H 2O (1) + 2NO 2(g)
C) 3Cu (s) + 8HNO3(aq) → 3Cu (NO3)2(aq) + 4H2O(1) + 2NO(g)
D) 3Cu(s) + 4HNO3(aq) → 3Cu(NO 3) 2(aq) + 2H 2O (1) + 2NO (g)
Show Answer
A) addition of water followed by filtration then sublimation
B) addition of water followed by sublimation then filtration
C) sublimation followed by addition of water then filtration
D) fractional distillation
E) fractional crystallization
Show Answer
Na2CO3(g) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)
How many moles of sodium trioxocarbonate (IV) are there in a 25 cm3 solution which required 10cm3 of 0.05 mol dm-3 hydrochloric acid solution to neutralize it?
Options:A) 1.000 mole
B) 0.100 mole
C) 0.010 mole`
D) 0.111 mole
Show Answer
0.0075 mole of calcium trioxocarbonate (IV) is added to 0.015 mole of a solution of hydrochloric acid. The volume of gas evolved at s.t.p is
[Molar volume of a gas at s.t.p = 22.4 dm3]
Options:A) 224cm3
B) 168cm3
C) 112cm3
D) 100cm3
Show Answer
A) It increase with increase in pressure
B) It increase at constant temperature
C) It increase with increase in temperature
D) It increase with increase in volume
Show Answer

2-methylbut-2-ene has the structure
Options:A) A
B) B
C) C
D) D
Show Answer
Beryllium and Aluminium have similar properties because they
Options:A) are both metals
B) belong to the same group
C) belong to the same period
D) are positioned diagonally to each other
Show Answer
A) 5 ml of (m/10) HCL
B) 10 ml of (m/10) HCL
C) 15 ml of (m/5) HCL
D) 20 ml of (m/8) HCL
Show Answer
A) Fe2+
B) Cl2
C) Fe3+
D) Cl-
E) H2O
Show Answer
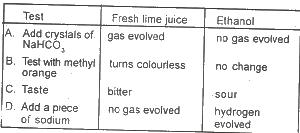
Use the option above to answer this question. Which is the correct set of results for tests conducted respectively on fresh lime juice and ethanol.
Options:A) A
B) B
C) C
D) D
Show Answer